Product Description
F1-X™ Next-Generation Gibson Assembly® Master Mix is an advanced cloning reagent developed to maximize efficiency and fidelity in seamless DNA assembly. This ready-to-use master mix enables single-tube cloning of 2-12 DNA fragments with exceptional performance on challenging multi-fragment constructs. Developed with guidance from Dr. Dan Gibson, inventor of Gibson Assembly®, F1-X™ represents a significant advancement in molecular cloning technology. The optimized enzyme blend achieves >95% cloning efficiency across all fragment counts, from simple 2-fragment joins to complex 12-fragment assemblies. Temperature flexibility (50-56°C) accommodates GC-rich and structured sequences that typically fail with standard cloning methods. Key advantages include 1-step workflow simplicity, compatibility with unpurified PCR products (≤20% v/v), cost-effective 10µL reaction volumes, and comprehensive quality control with included positive control DNA. Ideal applications include plasmid construction, metabolic pathway assembly, site-directed mutagenesis, combinatorial library generation, and synthetic biology projects requiring reliable multi-fragment cloning. Benchmarked directly against NEBuilder® HiFi across 2-, 7-, 9-, and 11-fragment assemblies, F1-X™ matched or exceeded competitor performance while reducing cost per reaction by 30%. Compatible with automated liquid handling systems for high-throughput cloning applications. 12-month shelf life at -20°C. For research use only. Not for use in diagnostic procedures.
Technical Details
| Reaction Conditions | |
| Temperature | 50°C (50–56°C range) |
| Time (2-3 fragments) | 15 min |
| Time (4-12 fragments) | 60 min |
| Reaction Volume | 20 µL standard (2.5 - 20 µL range) |
| DNA Input | 1-10 fmol/µL |
| Approx. ng/µL per fragment (20 µL rxn): 0.5 kb: 1–2 ng/µL · 1 kb: 0.5–1.25 ng/µL · 10 kb: 1.25–2.5 ng/µL | |
| Assembly Range | |
| Fragments | 2-12 per reaction |
| Fragment Size | 100 bp - 32 kb per fragment |
| Total Assembly Size | Up to 100 kb |
| Overlap (2-3 fragments) | 20-40 bp |
| Overlap (4+ fragments) | 40+ bp |
| Stability & Compatibility | |
| Storage | -20°C |
| Shelf Life | 12 months at -20°C (unopened) |
| Crude PCR Compatible | Yes (≤20% v/v) |
| Automation Compatible | Yes |
Workflow: Three Steps
Three steps take you from fragments to verified constructs with no special equipment
Step 1: Combine Fragments. Add fragments to F1-X™ Master Mix in a single tube.
Step 2: Incubate. 50°C for 15-60 minutes.
Step 3: Transform. Transform directly into competent cells or use cell-free workflows (PUREfrex®Cell-free Protein Synthesis).
F1-X Yielded Highest Average Error-free Clone Rate in a Head-to-Head Comparison of 1-step Assembly Mixes
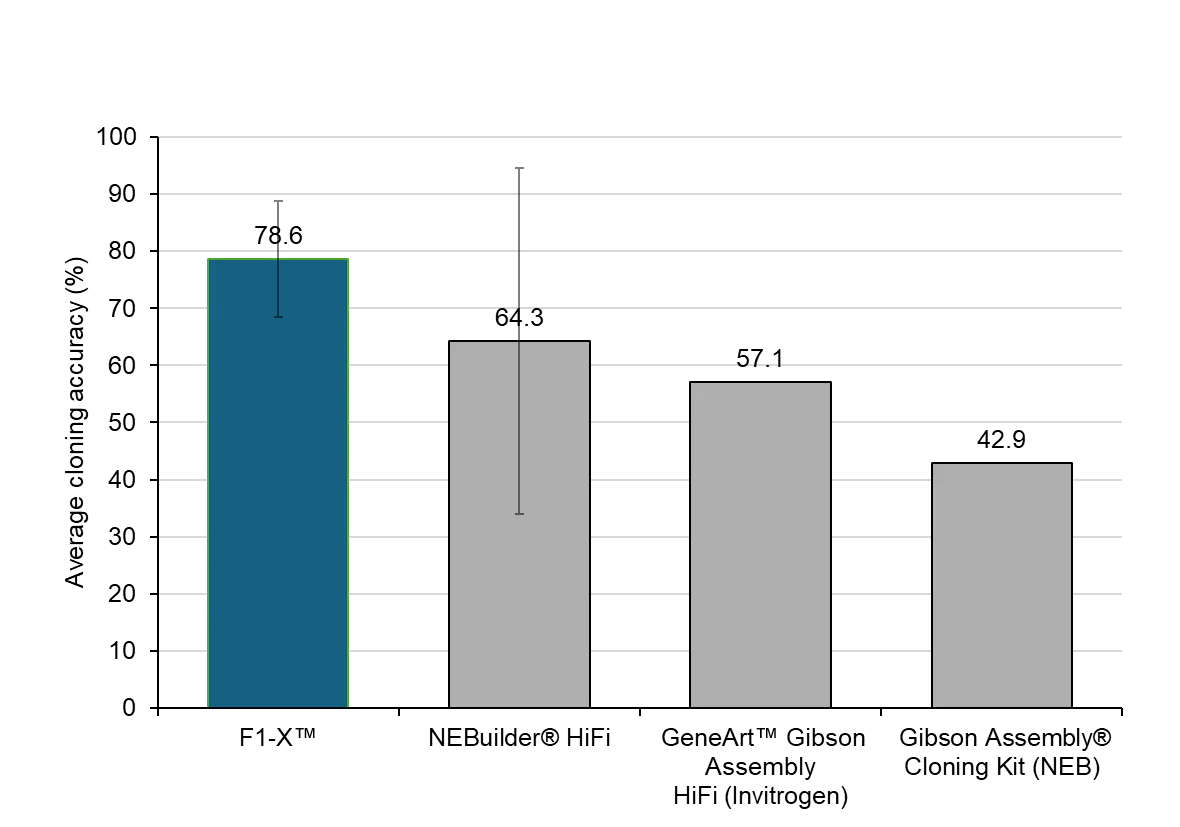
Head-to-head comparison of multi-fragment cloning accuracy. F1-X™ achieved 78.6% error-free clones, compared to 64.3% for NEBuilder® HiFi, 57.1% for GeneArt™ Gibson Assembly HiFi, and 42.9% for Gibson Assembly® Cloning Kit. In a 4-fragment assembly with 40 bp overlaps, F1-X™ exhibited the highest average cloning accuracy across the compared kits. Cloning accuracy was determined by whole-plasmid next-generation sequencing, with error-free clones defined as exhibiting 100% sequence identity to the reference and calculated as the fraction of error-free clones relative to total clones sequenced; for this analysis, cloning accuracy and error-free clone rate are used interchangeably. Full-length clones were pre-selected using a visual genetic reporter assay prior to Oxford Nanopore sequencing. Data represent n = 2 independent cloning and transformation experiments, with 7 colonies sequenced per replicate; error bars represent standard deviation between replicates. Cloning efficiency and fidelity are construct-dependent and may vary with sequence context and overlap design. Gibson Assembly® is a registered trademark of Telesis Bio Inc., used under license. NEBuilder® is a registered trademark of New England Biolabs, Inc. GeneArt™ is a trademark of Thermo Fisher Scientific.
Consistent Efficiency and Low Error Rates Across Increasing Assembly Complexity
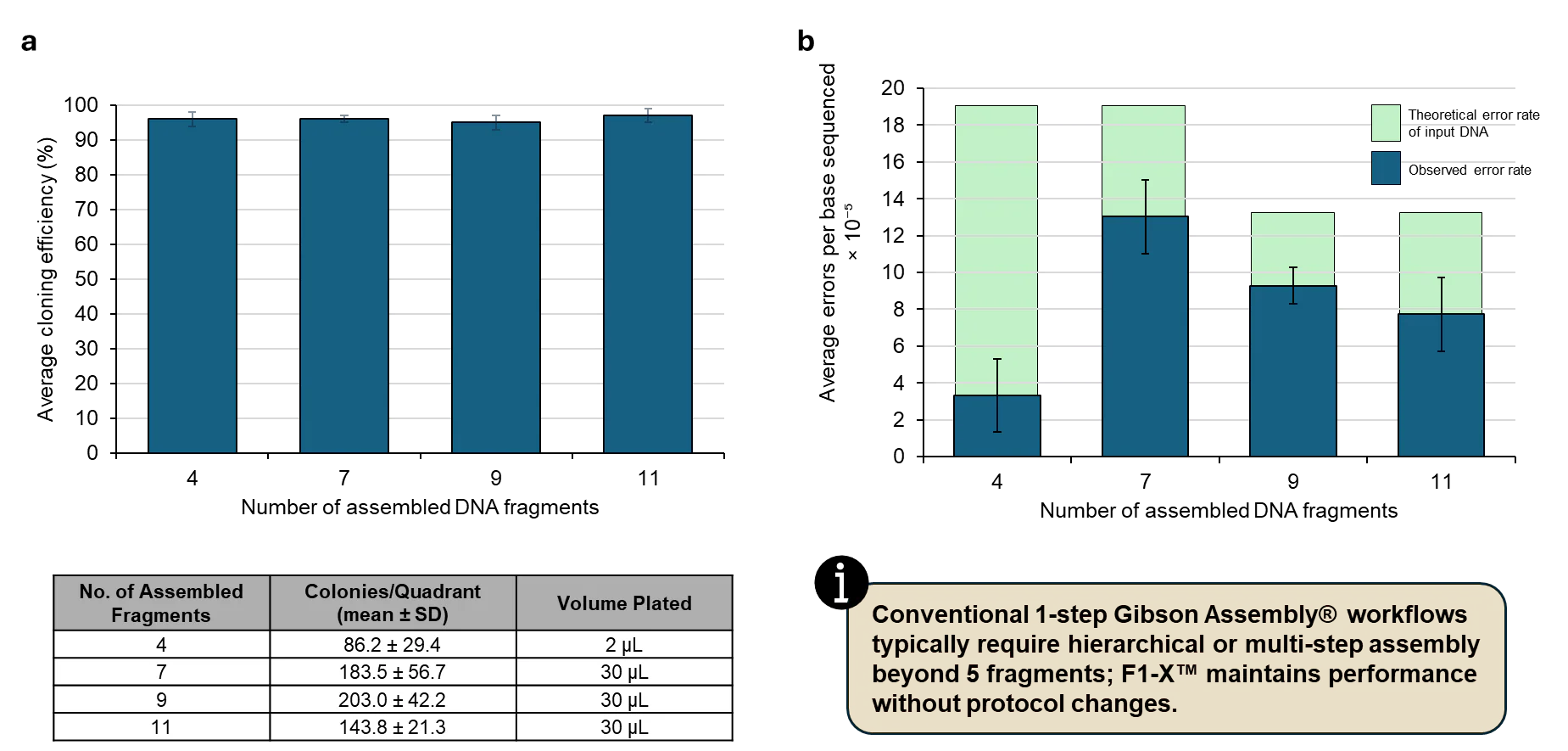
High performance and fidelity in multi-fragment cloning. F1-X™ delivers high cloning efficiency, robust colony counts, and high fidelity for complex cloning reactions in a simple, single assembly step—reducing or eliminating the need for multi-step assembly. Conventional Gibson Assembly methods may require hierarchical assembly or 2-step protocols to achieve acceptable efficiency beyond 4–5 fragments. In contrast, F1-X™ maintained 95-97% cloning efficiency and high fidelity across the tested complexities (4, 7, 9, and 11 fragments). Panel (a) shows the average cloning efficiency, determined using a fluorescent reporter system where clones containing the intended insert express a fluorescent reporter (efficiency = fluorescent colonies / total colonies). Colony counts for each reporter are also shown, demonstrating that ample colonies are formed for screening, even up to 11 fragments. Error bars represent the standard deviation of colony counts between n=4 agar plate quadrants. Panel (b) illustrates the high fidelity, which was measured by whole-plasmid Next-Generation Sequencing (NGS). The average error per base sequenced was calculated by the number of gaps, mismatches, or insertions in NGS consensus sequences divided by the total bases sequenced (construct length x number of clones sequenced). The theoretical error rate of the input DNA was estimated based on the preparation method: PCR of synthetic fragments for 4 and 7 fragment assemblies, and direct cloning of synthetic fragments for 9 and 11 fragment assemblies. Error bars represent the standard deviation between n=2 independent cloning reactions and transformations, with 7 colonies sequenced per replicate. 2/58 NGS consensus traces (3.4%) contained sequence gaps likely attributable to read quality or mapping artifacts and were excluded from fidelity analysis. The experimental approach was conducted as recommended in the F1-X User Guide. Fragments were designed with standard 40 bp overlaps, combined at equimolar ratios (0.02–0.04 pmol per fragment), and assembled under standard isothermal conditions (50°C, 1 hr). Synthetic DNA fragments (Twist Bioscience) were used directly or PCR-amplified prior to assembly. No optimization of fragment ratios, specialized overlap design, or multi-step assembly was required. For transformation, 1 µL of F1-X reaction was transformed into 20 µL chemically competent DH5α, rescued with SOC in 220 µL total volume, then plated. Note that cloning efficiency and fidelity are construct-dependent and may vary with sequence context and overlap design.
F1-X Maintains Performance in 11-fragment Assembly After Repeated Freeze-Thaw Stress
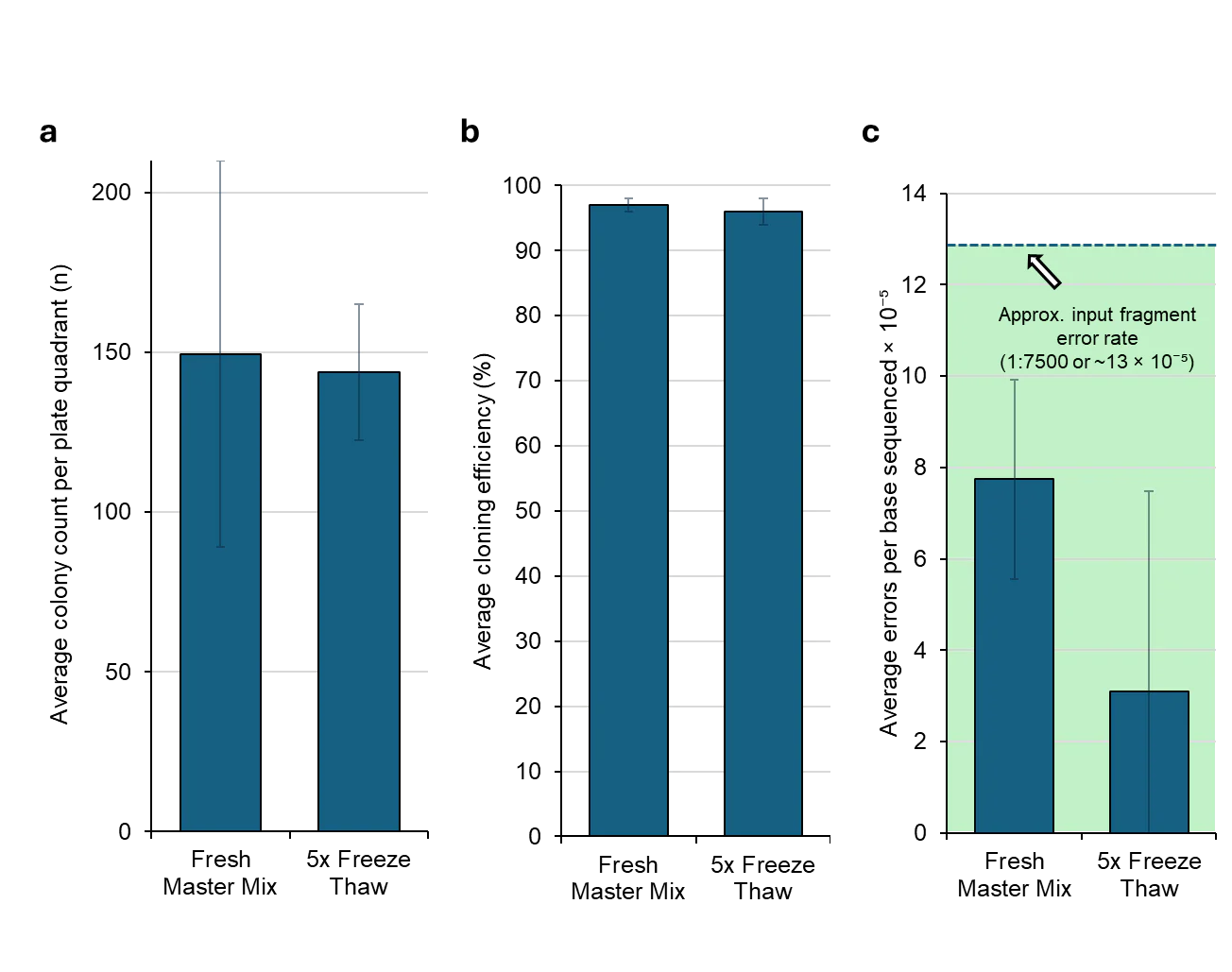
F1-X is robust to repeated freeze-thaw cycles. Many labs aliquot master mixes or experience unavoidable freeze-thaw events during routine use. In an 11-fragment assembly stress test, F1-X subjected to 5 freeze-thaw cycles showed no measurable decrease in (a) colony counts, (b) cloning efficiency (~95%), or (c) assembly fidelity compared with fresh master mix. Notably, observed error rates for both conditions (2.7–7.8 × 10⁻⁵ errors/bp) remained well below the published error rate specification for input synthetic DNA fragments (1:7,500 bp or ~13 × 10⁻⁵), confirming that F1-X assembly preserves input DNA fidelity rather than introducing additional errors. Notably, all inputs to the reaction were synthetic fragments directly from the synthesis plate. Dashed line indicates synthetic fragment error rate for reference. Note: due to its glycerol content, F1-X remains liquid at –20°C; however, cycling between –20°C and room temperature subjects the reagent to repeated thermal stress that mimics real-world storage and handling conditions. Experimental details are outlined below. Transformation: 1 µL reaction into 20 µL chemically competent DH5α, rescued in 220 µL total volume, 30 µL plated. n=2 replicates, 7 colonies sequenced per replicate; error bars in (c) represent the standard deviation between replicates. Error bars in (a) and (b) represent the standard deviation of colony counts in n=4 agar plate quadrants. Note that cloning efficiency and fidelity are construct-dependent and may vary with sequence context and overlap design.
F1-X Assembly Products Enable Transformation-free, Same-day Linear DNA Workflows
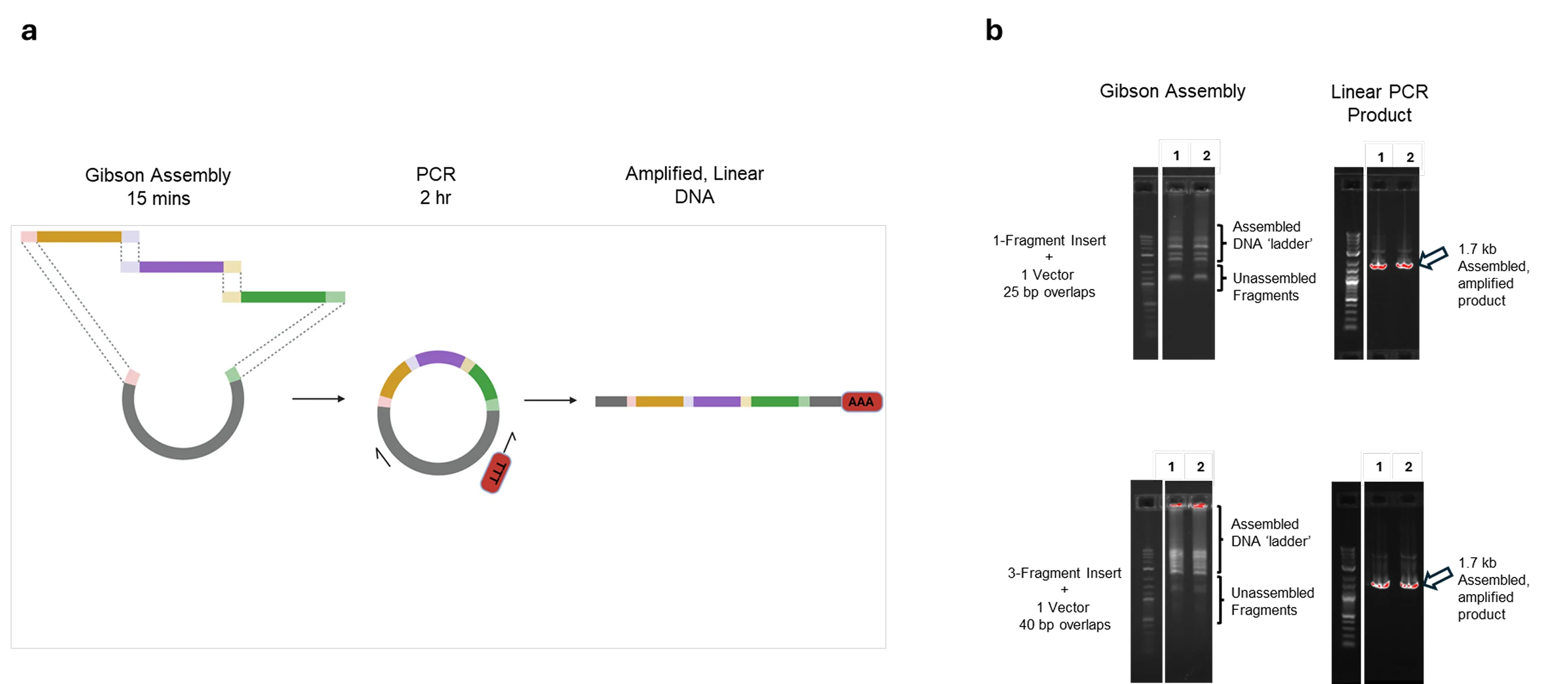
F1-X produces assembled products suitable for cell-free applications without transformation. For workflows requiring linear DNA templates such as in vitro transcription, cell-free protein expression, or direct PCR amplification, F1-X assembly products can be used directly without bacterial cloning. Panel (a) shows an overview of the assembly-to-amplification workflow. This approach is particularly useful for incorporating complex sequence elements such as poly(A) tails that are difficult to maintain in bacterial hosts. Panel (b) shows agarose gel electrophoresis of assembly products (left) and subsequent PCR amplification (right) for both simple (1-fragment insert + vector) and more complex (3-fragment insert + vector) assemblies. Lanes 1 and 2 represent independent replicates. Raw assembly products show the expected ladder of assembled intermediates and unassembled fragments; following PCR amplification across the assembled junction, both conditions resolved into a single, sharp 1.7 kb band of expected size, with no detectable mis-assembly or low molecular weight products. This indicates high ligation efficiency regardless of insert complexity. Assemblies were performed using standard F1-X recommended conditions and run for 15 minutes at 50°C. 1 µL of unpurified assembly product was added directly to a Platinum™ SuperFi™ II PCR reaction (standard reaction set up according to manufacturer recommendations) for amplification. All constructs were subsequently sequence-verified by NGS, confirming assembly fidelity. This streamlined workflow enables same-day generation of sequence-verified linear templates for applications where transformation is unnecessary or undesirable. Generally, we recommend a 15-minute assembly time for simple assemblies (≤3 fragments); for higher-complexity assemblies, a 60-minute reaction time is recommended as outlined in the F1-X User Guide. For this experiment, 15 minutes was sufficient to assemble both the 2- and 4-fragment constructs.
| Documents & Links for F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix | |
| Datasheet | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix User Guide |
| Application Note | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix Technical Application Note |
| Manual | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix Quick Start Guide |
| Documents & Links for F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix | |
| Datasheet | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix User Guide |
| Application Note | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix Technical Application Note |
| Manual | F1-X™ Next-Generation 1-Step Gibson Assembly® Master Mix Quick Start Guide |