Review: From Exosome Research to Extracellular Vesicle Analysis
Dr. Akira Yokoi
Japanese Society for Extracellular Vesicles Board Member
Department of Obstetrics and Gynecology, Nagoya University Hospital / Institute for Advanced Research
1. The Dawn of Exosome Research
The origins of exosome research date back to when small vesicles, approximately 100 nm in size, secreted by cells were first named "exosomes." Initially, exosomes were believed to be a mechanism for eliminating cellular waste1), and were not thought to play an active role in biological processes. However, in the late 1990s, it was reported that exosomes derived from immune cells could promote the maturation of antigen-presenting cells and were involved in immune responses2, 3). This shifted the perception of exosomes from mere byproducts to physiologically significant structures with specific functions. Then, in 2007, it was discovered that mRNA and microRNA within exosomes could be taken up by recipient cells, potentially altering gene expression4). This breakthrough was recognized globally as a "new mode of intercellular communication," marking a major leap forward in exosome research.
In the early days of research, 'exosome' became a widely used term, but it was later discovered that a variety of vesicles with different sizes and formation pathways were actually mixed together5). For example, microvesicles/ectosomes formed by outward budding of the cell membrane, and apoptotic bodies created during apoptosis, were also recognized as vesicles released outside the cell. These studies were conducted independently, yet they all shared the common idea that vesicles released from cells played a role in communication and maintaining homeostasis. It was standard practice to collectively call any pellet obtained by physical methods like ultracentrifugation or precipitation, regardless of its origin or formation route, an 'exosome.' As a result, the true nature and properties of the vesicles studied differed from report to report, making it difficult to compare results or verify reproducibility. However, as technology advanced such as with electron microscopy, nanoparticle tracking analysis (NTA), and proteomics, it became clear that extracellular vesicles are not a single uniform group, but rather are diverse in size, density, membrane composition, and secretion pathways. Recognizing this diversity became the foundation for establishing key concepts in later vesicle research and led to redefining the field within a broader context.
2. Establishing Extracellular Vesicles
With the rapid growth of exosome research, it became evident that membrane vesicles found outside cells are not a single homogeneous structure, but differ in their formation process and size. It became clear that exosomes were just one of several subtypes, a realization that prompted researchers to reconsider the terminology and definitions used in the field, revealing the limitations of centering everything around exosomes. In response to this evolving understanding, the International Society for Extracellular Vesicles (ISEV) was established in 2011, bringing together researchers from around the world. ISEV proposed the use of the term "extracellular vesicles (EVs)" as a standard to comprehensively describe membrane vesicles secreted outside the cell. EVs refer to all cell-derived, lipid bilayer-enclosed, non-replicating particles found outside cells, regardless of species, cell type, size, or isolation method. In this way, exosomes came to be recognized as one specific category within the broader group of EVs. The adoption of this comprehensive concept led to a recommendation that particles previously collectively reported as "exosomes" be distinguished based on their formation pathways and physical characteristics. Additionally, ISEV has worked to standardize reporting criteria for EV research, starting with MISEV2014 (Minimal Information for Studies of Extracellular Vesicles)6), followed by updates in 20187) and 20238). The latest MISEV2023 recommends distinguishing between EVs and non-vesicular extracellular particles (NVEPs) and clearly stating the “operational term” based on physical properties, cell of origin, and molecular contents. This standardized approach has improved reproducibility and comparability of research findings, and EV research is now advancing worldwide as a more rigorously defined scientific discipline.
3. The Foundations of Extracellular Vesicle Analysis
The shift in resolution from the broad perception of "exosome research" to more precise "extracellular vesicle (EV) analysis" is due to the realization that the EVs being analyzed are extremely diverse. Their size, origin, secretion pathways, and molecular cargo are not uniform, nor are the fractions obtained through various isolation, purification, or observation methods. Consequently, it is imperative for researchers to clearly define the properties of the EVs they handle during the analytical stage and to share this information in a comparable format. To this end, the MISEV guidelines are essential reading, as they serve to protect researchers when publishing their findings. Ensuring this fundamental transparency is the cornerstone of reliable EV research, which continues to be systematized based on the three pillars of reproducibility, standardization, and evaluation criteria.
It is both crucial and mandatory to clearly distinguish between EVs and NVEPs, recognize the selection bias inherent in various separation methods, and disclose the specific steps leading to functional analyses. These measures prevent the heterogeneity of EVs from being dismissed as “errors,” and this foundation is essential for treating EV diversity as a scientific concept rather than an anomaly and is vital for improving the comparability of studies. Additionally, moves toward standardized reporting formats and greater data sharing, supported by the development of electronic databases, are helping to promote international collaboration.
Advances in analytical technology have dramatically accelerated EV research. Techniques such as NTA and nano-flow cytometry for quantifying particle size and number, electron microscopy and cryo-tomography for structural analysis, and multi-omics profiling for comprehensive molecular characterization are just a few examples. Recently, single-EV analysis technologies have emerged, making it possible to directly measure proteins, nucleic acids, and glycans within individual EVs. Microfluidic devices and nanofiber materials now enable the capture and manipulation of EVs even in tiny sample volumes, allowing high-sensitivity detection. This fusion of diverse analytical modalities is shifting research from focusing on average EV characteristics to making differences among individuals, subtypes, and disease states more visible. As technology becomes more sophisticated, data complexity and interpretative challenges also increase. Even minor differences in separation methods can affect reproducibility, and the diversity of analytical systems can complicate comparisons between studies. To address this, researchers are encouraged to not only adopt innovative technologies but also critically assess the limitations and potential biases of their analytic systems and clearly document their research conditions. To truly understand EV diversity, it is essential to establish common benchmarks that are independent of specific methods or devices, and to organize data hierarchically by considering physical properties, molecular composition, and functional impact.
The primary goal of EV analysis research is not just to detect and identify particles, but to understand their biological significance within context. To answer questions regarding which EV subtypes are involved in specific physiological or pathological processes, or how secretion profiles shift in response to disease or environmental changes, an integrated approach combining both experimental and informatics methods is crucial. In disease-related EV studies, pinpointing which particles among the diverse EVs found in body fluids like blood and urine carry disease-specific information is achieved through "subtyping analysis," a key step in this field. Alongside this, efforts are underway to standardize EV analysis for clinical applications. When working with clinical samples, even slight differences in collection conditions, preservation methods, or isolation accuracy can impact results, highlighting the need for internationally shareable protocols. For diagnostic applications, it's important to identify the analyte, assess its homogeneity, and ensure reproducibility of the measurement environment. For therapeutic applications, quality indicators such as EV formulation characteristics, stability, and batch-to-batch consistency are essential. These efforts are clarifying the shift in perspective from viewing EVs as mere "analytes" to recognizing them as "functional units with biological activity."
In summary, the foundation of extracellular vesicle analysis goes beyond technical procedures; it is about embracing the diversity of EVs and establishing a scientific foundation that ensures research reproducibility and reliability. The combination of advancing analytical technologies, standardization, and a deep understanding of biological context will be fundamental pillars for the next generation of EV research.
4. Applications and the Future
Over the past decade, EV research has moved beyond the realm of basic biology and has expanded into fields such as disease biology, regenerative medicine, and drug discovery9). EVs reflect the state and environment of cells, and their molecular composition changes dynamically. EVs are increasingly recognized as vital biomarkers for disease diagnosis and for monitoring treatment efficacy. Recently, there has been growing interest in vesicles derived from plants, microbes, and even synthetic EVs created in the laboratory, extending beyond those originating solely from mammals. These studies suggest that EVs may represent a universal mechanism of intercellular communication across different species, taking our understanding of biological phenomena to a new level. A variety of technologies, including separation kits, flow cytometers for particle analysis, image analysis tools, and AI-driven data integration, are being adopted for EV analysis. Skillful use of these analytical tools to comprehensively examine EVs from molecular, structural, and functional perspectives will likely become the central focus of research in the coming decade. Through the integration of these advanced technologies, we can expect to gain more precise insights into the biological diversity and disease specificity of EVs, unlocking new functions and potential applications yet to be discovered. Ultimately, the continued advancement of EV research relies on both open collaboration and a return to scientific fundamentals. By integrating diverse expertise and striving for a precise scientific understanding of extracellular vesicles, we can ensure the reliability of applied research and its implementation in society. The future of EV research should be built on interdisciplinary co-creation and commitment to scientific rigor.
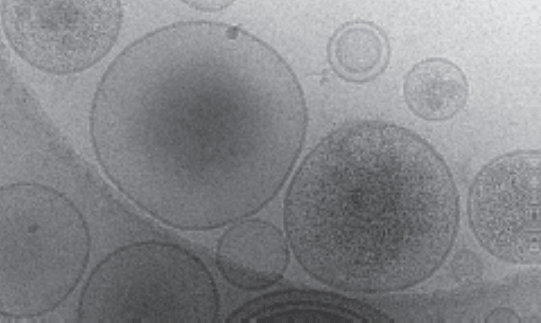
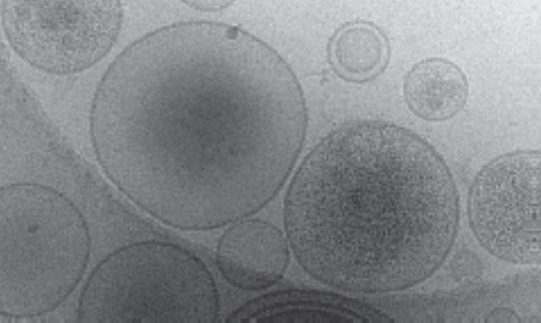 Figure: Heterogeneous EVs. Cryo-electron microscopy image of EVs extracted from human ovarian cancer cells. Even EVs extracted from a single clonal cell line exhibit heterogeneity in both size and their molecular cargo.
Figure: Heterogeneous EVs. Cryo-electron microscopy image of EVs extracted from human ovarian cancer cells. Even EVs extracted from a single clonal cell line exhibit heterogeneity in both size and their molecular cargo.References
1) Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C. The process of vesicle creation during reticulocyte maturation: Linking plasma membrane activities to released vesicles (exosomes). J Biol Chem. 1987;262(19):9412-20.
2) Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, et al. Eliminating established mouse tumors with a new cell-free vaccine: Exosomes produced by dendritic cells. Nat Med. 1998;4(5):594-600.
3) Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ, et al. B lymphocytes release vesicles that present antigens. J Exp Med. 1996;183(3):1161-72.
4) Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-based transfer of mRNAs and microRNAs: A novel pathway for genetic communication between cells. Nat Cell Biol. 2007;9(6):654-9.
5) Yokoi A, Ochiya T. Exosomes and extracellular vesicles: Re-evaluating their core roles in cancer biology. Semin Cancer Biol. 2021;74:79-91.
6) Lotvall J, Hill AF, Hochberg F, Buzas EI, Di Vizio D, Gardiner C, et al. Essential experimental standards for defining extracellular vesicles and their roles: A position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3:26913.
7) Thery C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750.
8) Welsh JA, Goberdhan DCI, O'Driscoll L, Buzas EI, Blenkiron C, Bussolati B, et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024;13(2):e12404.
9) Mizenko RR, Feaver M, Bozkurt BT, Lowe N, Nguyen B, Huang KW, et al. A critical systematic review of extracellular vesicle clinical trials. J Extracell Vesicles. 2024;13(10):e12510.
We would like to extend our heartfelt thanks to Dr. Akira Yokoi for contributing this manuscript.